Ingredients
Management
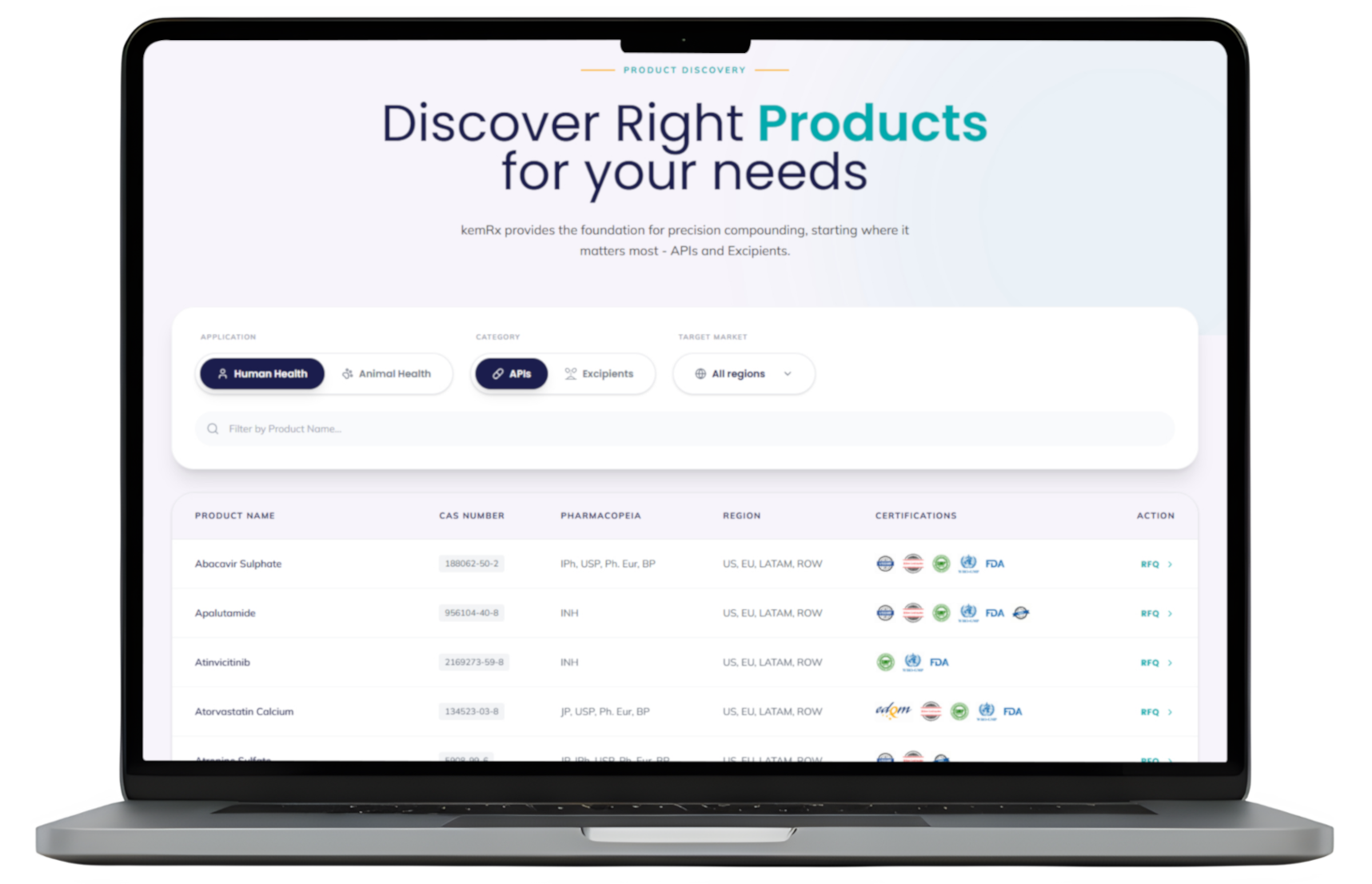
Centralized digital records for APIs and Excipients. Ensure quality, traceability, and regulatory integrity with our purpose-built material master data system.
APIs & Excipients

APIs
Sourced exclusively from GMP-certified manufacturers, our platform provides a comprehensive range of high-quality APIs across critical domains—including Weight Loss, Oncology, Cardiology, Diabetes, and Pain Management. We streamline your procurement process by integrating precise search capabilities with direct access to pharmacopeial grades and supplier certifications tailored to your target market.
- Human & Animal Health Grade
- Multi-compendial compliance
- Comprehensive Documentation Support
- Supply continuity

Excipients
Our rigorously selected portfolio of formulation-critical excipients—ranging from Tablet Coating Readymix and HPBCD to specialized grades of Crosspovidone (CR-L, M, S), HPC (LH 11, 21, 22), and PVP (K15 through K90)—is engineered to ensure absolute functional consistency and seamless compounding compatibility.
- Compendial excipients
- Functional coating systems
- Binders and diluents
- Ready-mix tablet coating materials
Engineered for Data Integrity
Material Master Data
Detailed profiles for every API and excipient. Structured critical data including CAS numbers, physical properties, and storage requirements.
Version-Controlled Documentation
Full lifecycle management. Track changes to specifications and Certified CoAs.
Change History & Traceability
Comprehensive audit trails of every batch movement and compliance check, ensuring 503A/503B readiness.